ONE-STOP MEDICAL DEVICE DEVELOPMENT COMPANY
FROM CONCEPT TO A WORLD-CLASS MEDTECH PRODUCT
Share your idea. As MedTech engineers we get it done!
What clients say about us?

 Since June 2022, we have been working with Consonance throughout the development process up to and including the BC-1 medical devices manufacturing. Their knowledge in the technical and regulatory field has been a huge gain. The agreed deliveries: time, quality and price have been delivered without deviation. We expect to continue our cooperation.
Since June 2022, we have been working with Consonance throughout the development process up to and including the BC-1 medical devices manufacturing. Their knowledge in the technical and regulatory field has been a huge gain. The agreed deliveries: time, quality and price have been delivered without deviation. We expect to continue our cooperation.
QA Manager
BrainCapture
Denmark
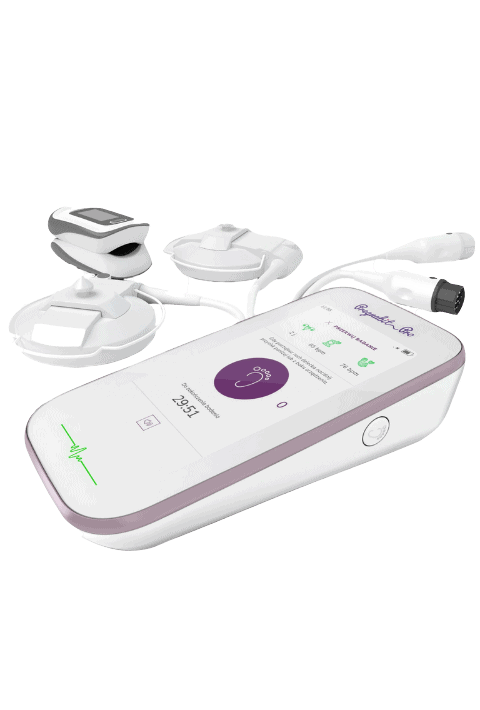
 “We worked together with Consonance to prepare functional prototypes of our impedance spectrometry and anal probe, the main parts of the ONIRY product. The cooperation was very good, the project was perfectly managed and communicated, all work went according to the schedule. We are fully satisfied.”
“We worked together with Consonance to prepare functional prototypes of our impedance spectrometry and anal probe, the main parts of the ONIRY product. The cooperation was very good, the project was perfectly managed and communicated, all work went according to the schedule. We are fully satisfied.”
PhD, CEO
OASIS Diagnostics
Poland
 "Consonance exhibited a high level of expertise, demonstrated exceptional organizational skills and a highly structured approach in conducting the feasibility study. The result they provided was not only thorough but also actionable, laying a solid foundation for the subsequent stages of the prototype development."
"Consonance exhibited a high level of expertise, demonstrated exceptional organizational skills and a highly structured approach in conducting the feasibility study. The result they provided was not only thorough but also actionable, laying a solid foundation for the subsequent stages of the prototype development."
MD, PhD
Charite
Germany
Our Clients
MedTech insights

From Diagnostics to AI – How Medical Devices Are Structured?
For clinicians, medical devices are tools that support diagnosis, treatment, and patient monitoring. But behind every interface and output lies a complex system that determines how reliable, interpretable, and clinically useful that device truly is.

We Build a New MedTech Alliance in Krakow
A new strategic MedTech alliance of the Faculty of Physics, Astronomy and Applied Computer Science, Centre for Technology Transfer CITTRU both from Jagiellonian University and Consonance has been formed in Krakow, marking an important step toward strengthening collaboration between industry and academia in the field of medical device development. “The partnership is built around a shared goal: accelerating the development of innovative MedTech solutions while equipping future professionals with the skills required to bring them to market. By combining Consonance’s hands-on medical product development and regulatory expertise with the university’s strong scientific and research background, both organizations aim to create a meaningful impact across the medical innovation ecosystem.” Driving Medical Device Development A core pillar of the collaboration is the joint execution of development projects focused on medical devices. These initiatives will span the full product lifecycle - from early-stage concept development and engineering to market readiness. The cooperation is designed to bridge the gap between theoretical research and real-world application, ensuring that promising ideas can be effectively translated into viable, compliant medical products. Building Practical MedTech Education Education will play a central role in the partnership. Experts from Consonance.tech will actively contribute to academic activities at Jagiellonian University, including lectures, workshops, and dedicated training sessions. The educational scope will cover key aspects of MedTech development, such as: product design and engineering processes, regulatory requirements and certification pathways, commercialization strategies in a highly regulated environment. This approach ensures that students and academic staff gain exposure to the practical realities of bringing medical technologies to market, going beyond purely theoretical knowledge. Creating Opportunities Through Internships To further support talent development, Consonance.tech will invite students and graduates of Jagiellonian University to a summer internship program. The program will offer direct involvement in ongoing MedTech projects, providing participants with hands-on experience in a multidisciplinary environment. Interns will have the opportunity to work alongside experienced engineers and project teams, gaining insights into real development processes, industry standards, and collaboration across technical and regulatory domains. Strengthening the Innovation Ecosystem This partnership reflects a broader commitment to fostering innovation in the MedTech sector. By enabling closer cooperation between academia and industry, Consonance and Jagiellonian University aim to support knowledge transfer, accelerate product development, and prepare the next generation of MedTech professionals. As the demand for advanced medical technologies continues to grow, initiatives like this play a critical role in building a sustainable and competitive innovation landscape - both in Poland and internationally.

Engineering Medical Innovations: Inside Consonance
Engineering medical innovations gave us two million medical devices across more than 7,000 categories (WHO), yet the demand for safer, smarter, and more innovative solutions never stops growing. Digital health technologies are transforming the way healthcare works, making it safer, more accessible, and more efficient. But behind every breakthrough device is an engineering team turning ideas into reality.