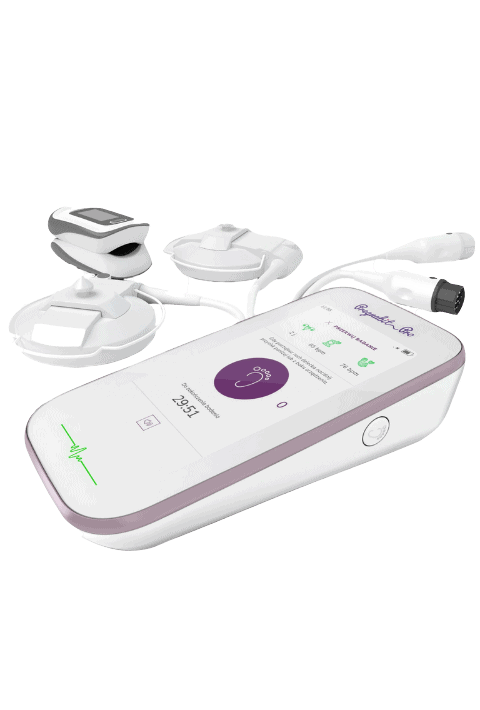
MEDTECH INNOVATIONS
Medical product design & development for pharma leaders
One-stop: we design, build and deliver efficiently at scale.
We partner pharmaceutical leaders and mid-cap healthcare companies.
Our goal is to build dependable medical products which create business value and improve people’s lives.
Diverse customer base and cooperation with leading universities and scientific institutes allows us to take a holistic approach to medical product development.
What clients say about us?

 Since June 2022, we have been working with Consonance throughout the development process up to and including the BC-1 medical devices manufacturing. Their knowledge in the technical and regulatory field has been a huge gain. The agreed deliveries: time, quality and price have been delivered without deviation. We expect to continue our cooperation.
Since June 2022, we have been working with Consonance throughout the development process up to and including the BC-1 medical devices manufacturing. Their knowledge in the technical and regulatory field has been a huge gain. The agreed deliveries: time, quality and price have been delivered without deviation. We expect to continue our cooperation.
Lisbeth Moller Christensen
QA Manager
BrainCapture
Denmark
QA Manager
BrainCapture
Denmark
 “We worked together with Consonance to prepare functional prototypes of our impedance spectrometry and anal probe, the main parts of the ONIRY product. The cooperation was very good, the project was perfectly managed and communicated, all work went according to the schedule. We are fully satisfied.”
“We worked together with Consonance to prepare functional prototypes of our impedance spectrometry and anal probe, the main parts of the ONIRY product. The cooperation was very good, the project was perfectly managed and communicated, all work went according to the schedule. We are fully satisfied.”
Katarzyna Borycka
PhD, CEO
OASIS Diagnostics
Poland
PhD, CEO
OASIS Diagnostics
Poland
 "Consonance exhibited a high level of expertise, demonstrated exceptional organizational skills and a highly structured approach in conducting the feasibility study. The result they provided was not only thorough but also actionable, laying a solid foundation for the subsequent stages of the prototype development."
"Consonance exhibited a high level of expertise, demonstrated exceptional organizational skills and a highly structured approach in conducting the feasibility study. The result they provided was not only thorough but also actionable, laying a solid foundation for the subsequent stages of the prototype development."
Melba Munoz
MD, PhD
Charite
Germany
MD, PhD
Charite
Germany
Our Clients
We help with

Developing better life-saving products
Process of creating innovative MedTech products means we always take into consideration the following factors: medical insights, business goals, market and user needs, features, unique differentiators and regulatory concerns.
Being bold is good in the conceptualization phase, but to build next gen sustainable medical devices we use proven processes and methods that help us deliver human-centered products that drive value.
Consonance’s medical product development lifecycle services get medical products to market efficiently at scale. We are ISO 13485 company and offer MDR certification support.
Let's talk about your next medical innovation!